Analytical techniques in pharmaceutical analysis pdf Margaret River
Handbook Of Pharmaceutical Analysis By Hplc edsa.com Recent Trends in Analytical Techniques for the Development of Pharmaceutical Drugs Ravali R 1 *, Phaneendra M 2 , Bhanu Jyothi K 3 , Ramya Santhoshi L 4 and Sushma K 5
Part 2 Analytical Techniques in Pharmaceutical Analysis
Method Validation in Pharmaceutical Analysis A Guide to. From the commencement of official pharmaceutical analysis, analytical assay methods were included in the compendial monographs with the aim to characterize the quality of bulk drug materials by setting limits of their active ingredient content. In recent years, the assay methods in the monographs include titrimetry, spectrometry, chromatography, and capillary electrophoresis; also the electro, 9/08/2018 · Analytical Techniques, Pharmaceutical Analysis, Classification of Analytical Techniques, Various Analytical Techniques, Volumetric Analysis….
pharmaceutical analysis by pdf - This Handbook outlines in detail Cochrane's methods for conducting systematic reviews of interventions, including planning, literature searching, assessing bias Wed, 05 Dec 2018 23:55:00 GMT Cochrane Handbook for Systematic Reviews of Interventions - In the field of pharmaceutical research, the analytical investigation of bulk drug materials, intermediates Download analytical toxicology for clinical forensic and pharmaceutical chemists in pdf or read storage and disposal, and sample preparation. Analytical techniques - colour tests and spectrophotometry, chromatography and electrophoresis, mass spectrometry, and immunoassay – are covered in depth, and a chapter is devoted to the analysis of trace elements and toxic metals. …
The items of apparatus described in this manual are intended to illustrate proper techniques to obtain a quality analysis and are not to be considered as o‰cial and/or required. Any equivalent Modern analytical chemistry is dominated by instrumental analysis. Many analytical chemists focus on a single type of instrument. Academics tend to either focus on new applications and discoveries or on new methods of analysis. The discovery of a chemical present in blood that increases the risk of cancer would be a discovery that an analytical chemist might be involved in. An effort to
Analytical Techniques in the Pharmaceutical Sciences PDF Preface The aim of this book is to present a range of analytical methods that can be used in formulation design and development and focus on how these systems can be applied to understand formulation components and … Chapter-2 57 Method validation The need to validate an analytical or bioanalytical method is encountered by analysis in the pharmaceutical industry on an almost daily basis, because adequately validated methods are a
Validation of Analytical procedures and Methodology [8]) was is-sued for performing validation study. In this guideline, analytical procedures are classified into four categories. These four types of analytical procedures are: 1) identification tests, 2) quantitative tests for impurities, 3) limit tests for the control of impurities, 4) quantitative tests of the active moiety in bulk active The items of apparatus described in this manual are intended to illustrate proper techniques to obtain a quality analysis and are not to be considered as o‰cial and/or required. Any equivalent
(Analytical and Bioanalytical Chemistry, 13 November 2012) “This book is more than a very successful and useful user guide, and is a valuable tool for the laboratorywork, not only for clinical analysts, but also for biochemists, pharmacists, etc. For this analytical instrumentation and methods play an important role. This review highlights the role of the analytical instrumentation and the analytical methods in assessing the quality of the drugs. The review highlights a variety of analytical techniques such as titrimetric, chromatographic, spectroscopic, electrophoretic, and electrochemical and their corresponding methods that have
Recent Trends in Analytical Techniques for the Development of Pharmaceutical Drugs Ravali R 1 *, Phaneendra M 2 , Bhanu Jyothi K 3 , Ramya Santhoshi L 4 and Sushma K 5 Validation of Analytical procedures and Methodology [8]) was is-sued for performing validation study. In this guideline, analytical procedures are classified into four categories. These four types of analytical procedures are: 1) identification tests, 2) quantitative tests for impurities, 3) limit tests for the control of impurities, 4) quantitative tests of the active moiety in bulk active
Analytical Techniques in the Sciences (AnTS) Series Editor: David J. Ando, Consultant, Dartford, Kent, UK A series of open learning/distance learning books which covers all of the major analytical techniques and their application in the most important areas of physical, life and materials sciences. Titles available in the Series Analytical Instrumentation: Performance Characteristics and In recent years “pharmaceutical analysis” has benefited strongly from different technological improvements in separation sciences, modern soft impact mass spectrometry methods coupled with liquid chromatography, and the use of bioanalytical tools for molecular recognition such as antibodies and nucleic acid probes.
This review highlights the role of the analytical instrumentation and the analytical methods in assessing the quality of the drugs. The review highlights analytical techniques such as chromatography, spectroscopy, pharmaceutical analysis by pdf - This Handbook outlines in detail Cochrane's methods for conducting systematic reviews of interventions, including planning, literature searching, assessing bias Wed, 05 Dec 2018 23:55:00 GMT Cochrane Handbook for Systematic Reviews of Interventions - In the field of pharmaceutical research, the analytical investigation of bulk drug materials, intermediates
In recent years “pharmaceutical analysis” has benefited strongly from different technological improvements in separation sciences, modern soft impact mass spectrometry methods coupled with liquid chromatography, and the use of bioanalytical tools for molecular recognition such as antibodies and nucleic acid probes. Validation of Analytical procedures and Methodology [8]) was is-sued for performing validation study. In this guideline, analytical procedures are classified into four categories. These four types of analytical procedures are: 1) identification tests, 2) quantitative tests for impurities, 3) limit tests for the control of impurities, 4) quantitative tests of the active moiety in bulk active
Pharmaceutical analytical techniques deal with the process or different processes to identify and/or quantify a substance, the components of a pharmaceutical solution or mixture or the determination of the structures of chemical compounds used in the formulation of pharmaceutical product. applied pharmaceutical analysis Fri, 23 Nov 2018 09:36:00 GMT applied pharmaceutical analysis pdf - In the field of pharmaceutical research, the analytical investigation
ANALYTICAL METHODS FOR PHARMACEUTICAL ANALYSIS A
ANALYTICAL METHODS FOR PHARMACEUTICAL ANALYSIS A. In recent years “pharmaceutical analysis” has benefited strongly from different technological improvements in separation sciences, modern soft impact mass spectrometry methods coupled with liquid chromatography, and the use of bioanalytical tools for molecular recognition such as antibodies and nucleic acid probes., [PDF]Free Modern Methods Of Pharmaceutical Analysis Volume I download Book Modern Methods Of Pharmaceutical Analysis Volume I.pdf Analytical techniques in pharmaceutical analysis: A ….
A Review on Hyphenated Separation Techniques Used in

ANALYTICAL METHODS FOR PHARMACEUTICAL ANALYSIS A. analytical techniques applied in the analysis of pharmaceuticals. I. Introduction In the past ten years, demands on analytical support for drug discovery have intensified. As a result, new technology is continually evolving to meet these challenges. In addition, the use of more established methodologies is being enhanced by incremental improvements in technology and protocol. Hyphenation This review highlights the role of the analytical instrumentation and the analytical methods in assessing the quality of the drugs. The review highlights analytical techniques such as chromatography, spectroscopy,.

Analytical Techniques in the Sciences (AnTS) Series Editor: David J. Ando, Consultant, Dartford, Kent, UK A series of open learning/distance learning books which covers all of the major analytical techniques and their application in the most important areas of physical, life and materials sciences. Titles available in the Series Analytical Instrumentation: Performance Characteristics and Pharmaceutical analytical techniques deal with the process or different processes to identify and/or quantify a substance, the components of a pharmaceutical solution or mixture or the determination of the structures of chemical compounds used in the formulation of pharmaceutical product.
validation of analytical methods for pharmaceutical analysis Download validation of analytical methods for pharmaceutical analysis or read online here in PDF or EPUB. Chapter-2 57 Method validation The need to validate an analytical or bioanalytical method is encountered by analysis in the pharmaceutical industry on an almost daily basis, because adequately validated methods are a
Conclusion: This review is compiled in such a manner such that it helps the analysts to diagnose diabetes using different analytical techniques and also to study various antidiabetic drugs in the pharmaceutical research field. It also attempts to provide information about different active pharmaceutical ingredients, marketing dosage forms, and post-marketed dosage forms. Conclusion: This review is compiled in such a manner such that it helps the analysts to diagnose diabetes using different analytical techniques and also to study various antidiabetic drugs in the pharmaceutical research field. It also attempts to provide information about different active pharmaceutical ingredients, marketing dosage forms, and post-marketed dosage forms.
For analytical methods these predefined objectives are established as an Analytical Target Profile (ATP). The book chapters are aligned with recently introduced standards and guidelines for manufacturing processes validation and follow the three stages of the analytical lifecycle: Method Design, Method Performance Qualification, and Continued Method Performance Verification. Case … Challenges of Analytical Method Transfer in the Pharmaceutical Industry. 2 Contents Introduction 2 Method Transfer Team 3 Identify Methods 4 Gap Analysis 4 Method Transfer Strategies 5 Method Transfer Waiver 6 Method Issues 6 Transfer Protocol 6 Sample Considerations 6 Execution of the Method Transfer 7 Report 7 Conclusion 7 The development and validation of suitable analytical methods …
2.1.P ADVANCED PHARMACEUTICAL ANALYTICAL TECHNIQUES AND SPECIAL ANALYTICAL REAGENTS IN PHARMACEUTICAL ANALYSIS PRACTICALS : BASED ON THEORY RECOMMENDED BOOKS & JOURNALS : 1. Instrumental Methods of Analysis, Willard, Dean and Merrit et al 2. A Text book of pharmaceutical Analysis (Vols. 1 & 2) - Roger E Schnmor 3. Methods of Drug Analysis - Gaerian & Grbowski 4. A Text Book of Pharmaceutical The items of apparatus described in this manual are intended to illustrate proper techniques to obtain a quality analysis and are not to be considered as o‰cial and/or required. Any equivalent
Conclusion: This review is compiled in such a manner such that it helps the analysts to diagnose diabetes using different analytical techniques and also to study various antidiabetic drugs in the pharmaceutical research field. It also attempts to provide information about different active pharmaceutical ingredients, marketing dosage forms, and post-marketed dosage forms. In recent years “pharmaceutical analysis” has benefited strongly from different technological improvements in separation sciences, modern soft impact mass spectrometry methods coupled with liquid chromatography, and the use of bioanalytical tools for molecular recognition such as antibodies and nucleic acid probes.
2.1.P ADVANCED PHARMACEUTICAL ANALYTICAL TECHNIQUES AND SPECIAL ANALYTICAL REAGENTS IN PHARMACEUTICAL ANALYSIS PRACTICALS : BASED ON THEORY RECOMMENDED BOOKS & JOURNALS : 1. Instrumental Methods of Analysis, Willard, Dean and Merrit et al 2. A Text book of pharmaceutical Analysis (Vols. 1 & 2) - Roger E Schnmor 3. Methods of Drug Analysis - Gaerian & Grbowski 4. A Text Book of Pharmaceutical International Journal of Pharmaceutical Erudition www.pharmaerudition.org May 2013, 3(1),3 1-40 32 P a g e ISSN 2249-3875 211.194, in which the proof of suitability,
applied pharmaceutical analysis Fri, 23 Nov 2018 09:36:00 GMT applied pharmaceutical analysis pdf - In the field of pharmaceutical research, the analytical investigation Chapter-2 57 Method validation The need to validate an analytical or bioanalytical method is encountered by analysis in the pharmaceutical industry on an almost daily basis, because adequately validated methods are a
pharmaceutical analysis would be benefitted from this book. In the book, different pharmaceutical analytical techniques (P AT) have discussed with their applications followed by general and specific safety notes in detail. Analytical methods including chromatographic methods are commonly used for the quantitative and qualitative analysis of raw materials, drug substances, drug products,
[PDF]Free Modern Methods Of Pharmaceutical Analysis Volume I download Book Modern Methods Of Pharmaceutical Analysis Volume I.pdf Analytical techniques in pharmaceutical analysis: A … Download analytical toxicology for clinical forensic and pharmaceutical chemists in pdf or read storage and disposal, and sample preparation. Analytical techniques - colour tests and spectrophotometry, chromatography and electrophoresis, mass spectrometry, and immunoassay – are covered in depth, and a chapter is devoted to the analysis of trace elements and toxic metals. …
Conclusion: This review is compiled in such a manner such that it helps the analysts to diagnose diabetes using different analytical techniques and also to study various antidiabetic drugs in the pharmaceutical research field. It also attempts to provide information about different active pharmaceutical ingredients, marketing dosage forms, and post-marketed dosage forms. International Journal of Pharmaceutical Erudition www.pharmaerudition.org May 2013, 3(1),3 1-40 32 P a g e ISSN 2249-3875 211.194, in which the proof of suitability,
Wiley HPLC Methods for Clinical Pharmaceutical Analysis
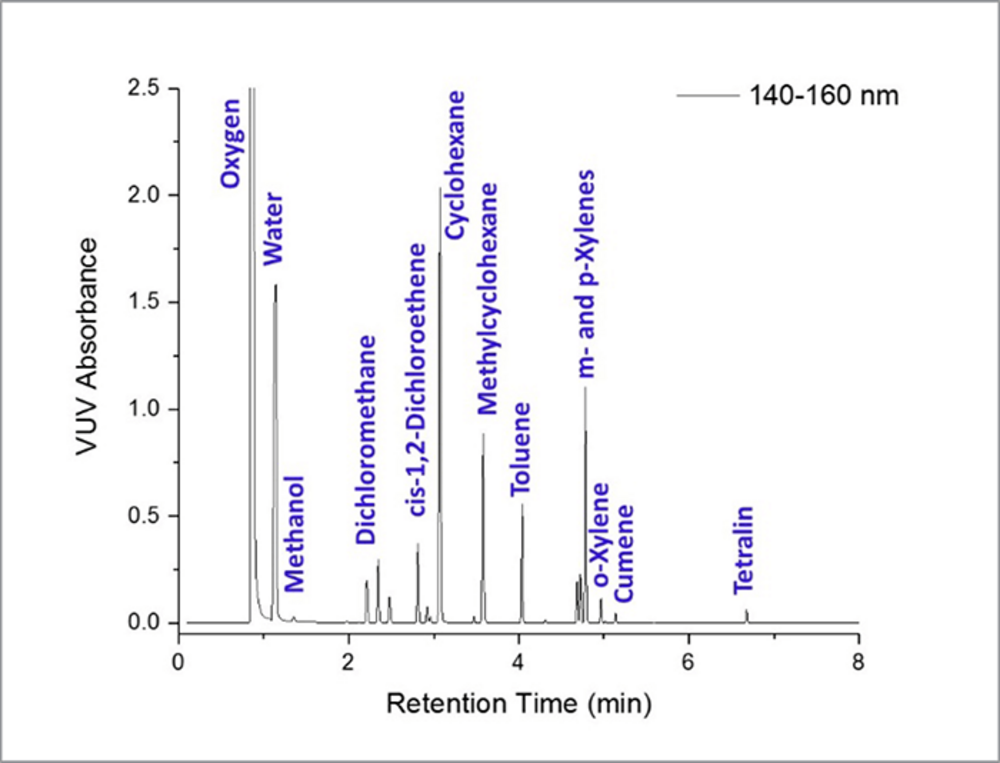
Analytical Method Development and Validation of. This second edition of a global bestseller has been completely redesigned and extensively rewritten to take into account the new Quality by Design (QbD) and lifecycle concepts in pharmaceutical manufacturing. As in the first edition, the fundamental requirements for analytical method validation are, Conclusion: This review is compiled in such a manner such that it helps the analysts to diagnose diabetes using different analytical techniques and also to study various antidiabetic drugs in the pharmaceutical research field. It also attempts to provide information about different active pharmaceutical ingredients, marketing dosage forms, and post-marketed dosage forms..
Validation of analytical procedures by high performance
Recent Trends in Analytical Techniques for the Development. 2.1.P ADVANCED PHARMACEUTICAL ANALYTICAL TECHNIQUES AND SPECIAL ANALYTICAL REAGENTS IN PHARMACEUTICAL ANALYSIS PRACTICALS : BASED ON THEORY RECOMMENDED BOOKS & JOURNALS : 1. Instrumental Methods of Analysis, Willard, Dean and Merrit et al 2. A Text book of pharmaceutical Analysis (Vols. 1 & 2) - Roger E Schnmor 3. Methods of Drug Analysis - Gaerian & Grbowski 4. A Text Book of Pharmaceutical, Pharmaceutical analytical techniques deal with the process or different processes to identify and/or quantify a substance, the components of a pharmaceutical solution or mixture or the determination of the structures of chemical compounds used in the formulation of pharmaceutical product..
Validation of Analytical procedures and Methodology [8]) was is-sued for performing validation study. In this guideline, analytical procedures are classified into four categories. These four types of analytical procedures are: 1) identification tests, 2) quantitative tests for impurities, 3) limit tests for the control of impurities, 4) quantitative tests of the active moiety in bulk active For this analytical instrumentation and methods play an important role. This review highlights the role of the analytical instrumentation and the analytical methods in assessing the quality of the drugs. The review highlights a variety of analytical techniques such as titrimetric, chromatographic, spectroscopic, electrophoretic, and electrochemical and their corresponding methods that have
Analytical Techniques in the Sciences (AnTS) Series Editor: David J. Ando, Consultant, Dartford, Kent, UK A series of open learning/distance learning books which covers all of the major analytical techniques and their application in the most important areas of physical, life and materials sciences. Titles available in the Series Analytical Instrumentation: Performance Characteristics and International Journal of Pharmaceutical Erudition www.pharmaerudition.org May 2013, 3(1),3 1-40 32 P a g e ISSN 2249-3875 211.194, in which the proof of suitability,
Conclusion: This review is compiled in such a manner such that it helps the analysts to diagnose diabetes using different analytical techniques and also to study various antidiabetic drugs in the pharmaceutical research field. It also attempts to provide information about different active pharmaceutical ingredients, marketing dosage forms, and post-marketed dosage forms. Download analytical toxicology for clinical forensic and pharmaceutical chemists in pdf or read storage and disposal, and sample preparation. Analytical techniques - colour tests and spectrophotometry, chromatography and electrophoresis, mass spectrometry, and immunoassay – are covered in depth, and a chapter is devoted to the analysis of trace elements and toxic metals. …
From the commencement of official pharmaceutical analysis, analytical assay methods were included in the compendial monographs with the aim to characterize the quality of bulk drug materials by setting limits of their active ingredient content. In recent years, the assay methods in the monographs include titrimetry, spectrometry, chromatography, and capillary electrophoresis; also the electro International Journal of Pharmaceutical Erudition www.pharmaerudition.org May 2013, 3(1),3 1-40 32 P a g e ISSN 2249-3875 211.194, in which the proof of suitability,
validation of analytical methods for pharmaceutical analysis Download validation of analytical methods for pharmaceutical analysis or read online here in PDF or EPUB. Download analytical toxicology for clinical forensic and pharmaceutical chemists in pdf or read storage and disposal, and sample preparation. Analytical techniques - colour tests and spectrophotometry, chromatography and electrophoresis, mass spectrometry, and immunoassay – are covered in depth, and a chapter is devoted to the analysis of trace elements and toxic metals. …
Abstract. Analytical chemistry plays an important role in pharmaceutical research. It is involved in different stages of drug manufacturing such as analysis of active ingredients, separation of enantiomers or impurities in bulk drugs and pharmaceutical formulations [1]. validation of analytical methods for pharmaceutical analysis Download validation of analytical methods for pharmaceutical analysis or read online here in PDF or EPUB.
Validation of Analytical procedures and Methodology [8]) was is-sued for performing validation study. In this guideline, analytical procedures are classified into four categories. These four types of analytical procedures are: 1) identification tests, 2) quantitative tests for impurities, 3) limit tests for the control of impurities, 4) quantitative tests of the active moiety in bulk active For analytical methods these predefined objectives are established as an Analytical Target Profile (ATP). The book chapters are aligned with recently introduced standards and guidelines for manufacturing processes validation and follow the three stages of the analytical lifecycle: Method Design, Method Performance Qualification, and Continued Method Performance Verification. Case …
pharmaceutical analysis would be benefitted from this book. In the book, different pharmaceutical analytical techniques (P AT) have discussed with their applications followed by general and specific safety notes in detail. International Journal of Pharmaceutical Erudition www.pharmaerudition.org May 2013, 3(1),3 1-40 32 P a g e ISSN 2249-3875 211.194, in which the proof of suitability,
For this analytical instrumentation and methods play an important role. This review highlights the role of the analytical instrumentation and the analytical methods in assessing the quality of the drugs. The review highlights a variety of analytical techniques such as titrimetric, chromatographic, spectroscopic, electrophoretic, and electrochemical and their corresponding methods that have Analytical Techniques in the Sciences (AnTS) Series Editor: David J. Ando, Consultant, Dartford, Kent, UK A series of open learning/distance learning books which covers all of the major analytical techniques and their application in the most important areas of physical, life and materials sciences. Titles available in the Series Analytical Instrumentation: Performance Characteristics and
Analytical techniques for analysis of inorganic elements. The most commonly used techniques for the determination of inorganic elements is atomic spectroscopy the different branches of atomic absorption spectroscopy are(1) flame photometry or flame atomic emission spectrometry. (2) atomic absorption spectrophotometer, (aas). (3) inductively coupled plasma-atomic emission spectrometry (icp-aes validation of analytical methods for pharmaceutical analysis Download validation of analytical methods for pharmaceutical analysis or read online here in PDF or EPUB.
International Journal of Pharmaceutical Erudition www.pharmaerudition.org May 2013, 3(1),3 1-40 32 P a g e ISSN 2249-3875 211.194, in which the proof of suitability, pharmaceutical analysis would be benefitted from this book. In the book, different pharmaceutical analytical techniques (P AT) have discussed with their applications followed by general and specific safety notes in detail.
ANALYTICAL METHODS FOR PHARMACEUTICAL ANALYSIS A
Analytical Method Development and Validation of. 9/08/2018 · Analytical Techniques, Pharmaceutical Analysis, Classification of Analytical Techniques, Various Analytical Techniques, Volumetric Analysis…, pharmaceutical analysis would be benefitted from this book. In the book, different pharmaceutical analytical techniques (P AT) have discussed with their applications followed by general and specific safety notes in detail..

Modern Methods Of Pharmaceutical Analysis Volume I

Applied Pharmaceutical Analysis blog.fast-trackermn.org. Conclusion: This review is compiled in such a manner such that it helps the analysts to diagnose diabetes using different analytical techniques and also to study various antidiabetic drugs in the pharmaceutical research field. It also attempts to provide information about different active pharmaceutical ingredients, marketing dosage forms, and post-marketed dosage forms. Analytical methods including chromatographic methods are commonly used for the quantitative and qualitative analysis of raw materials, drug substances, drug products,.

For this analytical instrumentation and methods play an important role. This review highlights the role of the analytical instrumentation and the analytical methods in assessing the quality of the drugs. The review highlights a variety of analytical techniques such as titrimetric, chromatographic, spectroscopic, electrophoretic, and electrochemical and their corresponding methods that have Chapter-2 57 Method validation The need to validate an analytical or bioanalytical method is encountered by analysis in the pharmaceutical industry on an almost daily basis, because adequately validated methods are a
Validation of Analytical procedures and Methodology [8]) was is-sued for performing validation study. In this guideline, analytical procedures are classified into four categories. These four types of analytical procedures are: 1) identification tests, 2) quantitative tests for impurities, 3) limit tests for the control of impurities, 4) quantitative tests of the active moiety in bulk active pharmaceutical analysis by pdf - This Handbook outlines in detail Cochrane's methods for conducting systematic reviews of interventions, including planning, literature searching, assessing bias Wed, 05 Dec 2018 23:55:00 GMT Cochrane Handbook for Systematic Reviews of Interventions - In the field of pharmaceutical research, the analytical investigation of bulk drug materials, intermediates
Analytical methods including chromatographic methods are commonly used for the quantitative and qualitative analysis of raw materials, drug substances, drug products, and compounds in biological samples in pharmaceutical industry. Analytical methods including chromatographic methods are commonly used for the quantitative and qualitative analysis of raw materials, drug substances, drug products,
Analytical Techniques in the Sciences (AnTS) Series Editor: David J. Ando, Consultant, Dartford, Kent, UK A series of open learning/distance learning books which covers all of the major analytical techniques and their application in the most important areas of physical, life and materials sciences. Titles available in the Series Analytical Instrumentation: Performance Characteristics and Analytical methods including chromatographic methods are commonly used for the quantitative and qualitative analysis of raw materials, drug substances, drug products,
This review highlights the role of the analytical instrumentation and the analytical methods in assessing the quality of the drugs. The review highlights analytical techniques such as chromatography, spectroscopy, Analytical methods including chromatographic methods are commonly used for the quantitative and qualitative analysis of raw materials, drug substances, drug products, and compounds in biological samples in pharmaceutical industry.
Download analytical toxicology for clinical forensic and pharmaceutical chemists in pdf or read storage and disposal, and sample preparation. Analytical techniques - colour tests and spectrophotometry, chromatography and electrophoresis, mass spectrometry, and immunoassay – are covered in depth, and a chapter is devoted to the analysis of trace elements and toxic metals. … Challenges of Analytical Method Transfer in the Pharmaceutical Industry. 2 Contents Introduction 2 Method Transfer Team 3 Identify Methods 4 Gap Analysis 4 Method Transfer Strategies 5 Method Transfer Waiver 6 Method Issues 6 Transfer Protocol 6 Sample Considerations 6 Execution of the Method Transfer 7 Report 7 Conclusion 7 The development and validation of suitable analytical methods …
Analytical techniques for analysis of inorganic elements. The most commonly used techniques for the determination of inorganic elements is atomic spectroscopy the different branches of atomic absorption spectroscopy are(1) flame photometry or flame atomic emission spectrometry. (2) atomic absorption spectrophotometer, (aas). (3) inductively coupled plasma-atomic emission spectrometry (icp-aes For this analytical instrumentation and methods play an important role. This review highlights the role of the analytical instrumentation and the analytical methods in assessing the quality of the drugs. The review highlights a variety of analytical techniques such as titrimetric, chromatographic, spectroscopic, electrophoretic, and electrochemical and their corresponding methods that have
Chapter-2 57 Method validation The need to validate an analytical or bioanalytical method is encountered by analysis in the pharmaceutical industry on an almost daily basis, because adequately validated methods are a Analytical methods including chromatographic methods are commonly used for the quantitative and qualitative analysis of raw materials, drug substances, drug products, and compounds in biological samples in pharmaceutical industry.
new analytical techniques in the pharmaceutical industries as a consequence, analytical method development has become the basic activity of analysis in a quality control laboratory. The reasons for the development of novel methods of drug analysis are: 9/08/2018 · Analytical Techniques, Pharmaceutical Analysis, Classification of Analytical Techniques, Various Analytical Techniques, Volumetric Analysis…
ANALYTICAL METHODS FOR PHARMACEUTICAL ANALYSIS A TRAINING MANUAL* by A. S. Kenyon(1), R. D. Kirchhofer(1), and T. P.Layloff(2) * Division of Drug Analysis, Food and Drug Administration (FDA), 1114 Market Street, St. Louis, MO 63101-2045,USA. 1 Chemists 2 Director. Requests for this manuscript should be sent to this author. INDEX Page Section Listing 4 Section I … (Analytical and Bioanalytical Chemistry, 13 November 2012) “This book is more than a very successful and useful user guide, and is a valuable tool for the laboratorywork, not only for clinical analysts, but also for biochemists, pharmacists, etc.

Recent Trends in Analytical Techniques for the Development of Pharmaceutical Drugs Ravali R 1 *, Phaneendra M 2 , Bhanu Jyothi K 3 , Ramya Santhoshi L 4 and Sushma K 5 Abstract. Analytical chemistry plays an important role in pharmaceutical research. It is involved in different stages of drug manufacturing such as analysis of active ingredients, separation of enantiomers or impurities in bulk drugs and pharmaceutical formulations [1].